CRISPR gene editing is rapidly transforming the future of medicine, offering precise and powerful ways to correct genetic defects at their source. From curing inherited diseases to fighting cancer and customizing treatments based on your DNA, CRISPR is leading a revolution in healthcare. In this blog post, we explore how this groundbreaking technology works, its real-world success stories, ethical challenges, and bold predictions for 2030—revealing why CRISPR may soon become the foundation of modern human health.
What Is CRISPR? A Simple Explanation of the Science Behind Gene Editing
CRISPR gene editing is a groundbreaking scientific method that allows scientists to precisely alter DNA within living organisms. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats, which are natural sequences found in the DNA of bacteria. These sequences help bacteria defend themselves against viruses, and researchers have adapted this system into a powerful tool for cutting and modifying genes in humans, animals, and plants.
At the core of CRISPR gene editing is a protein called Cas9, which acts like molecular scissors. Scientists program Cas9 with a piece of guide RNA that matches a specific DNA sequence. When introduced into a cell, Cas9 locates the target gene and makes a clean cut, allowing researchers to remove, repair, or replace the genetic material. This technology is faster, more accurate, and less expensive than older gene-editing methods, making it a revolutionary step toward treating genetic diseases and customizing medical treatments.
The Evolution of Gene Editing: From Discovery to 2030 Vision
The journey of gene editing began in the 1970s with early experiments in recombinant DNA, but true breakthroughs came decades later. The discovery of CRISPR gene editing in 2012 by scientists Jennifer Doudna and Emmanuelle Charpentier marked a turning point, offering a faster, more precise, and cost-effective way to alter DNA. Since then, CRISPR has advanced from lab research to real-world medical applications, including promising clinical trials for diseases like sickle cell anemia and certain cancers.
As we look ahead to 2030, CRISPR gene editing is expected to become a core tool in personalized medicine. Experts predict that treatments for previously untreatable genetic disorders will become more common, and CRISPR could even be used preventively to stop diseases before symptoms appear. With ongoing improvements in accuracy, reduced side effects, and expanded global access, the next five years may see CRISPR evolve from an experimental breakthrough to a routine part of human healthcare.
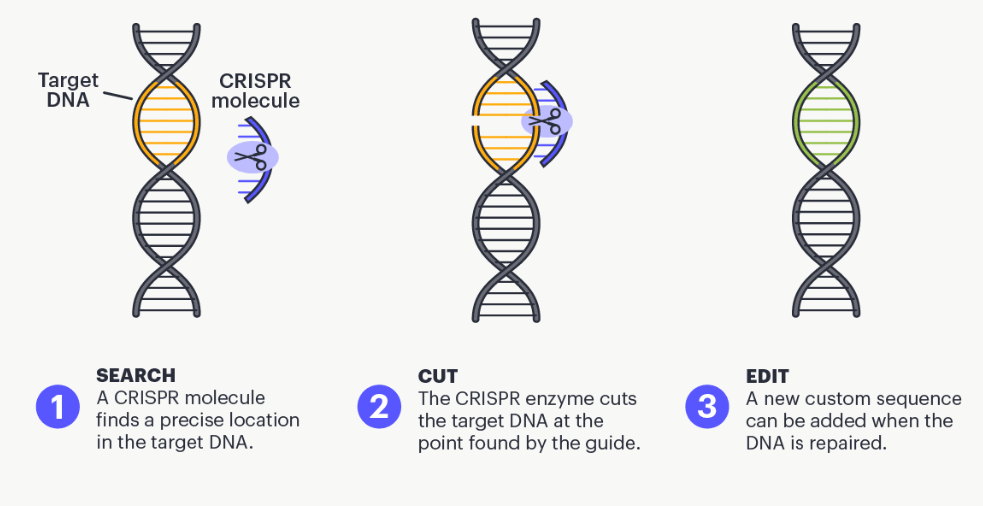
How CRISPR Gene Editing Works at the Molecular Level
CRISPR gene editing works like a precise molecular scalpel that can target and modify specific parts of DNA. At the heart of this system is a protein called Cas9 and a piece of guide RNA (gRNA). The guide RNA is designed to match the DNA sequence scientists want to edit. Once introduced into a cell, the guide RNA leads the Cas9 protein to the exact location in the genome that needs to be changed.
When Cas9 reaches the target DNA, it cuts both strands of the DNA helix at the specified spot. This break activates the cell’s natural repair process. Scientists can use this moment to either disable a faulty gene, correct a mutation, or insert new genetic material. CRISPR gene editing is so powerful because it offers high precision, low cost, and versatility—making it one of the most promising tools in modern molecular biology and medicine.
CRISPR vs. Traditional Genetic Treatments: What’s the Difference?
Traditional genetic treatments, such as gene therapy, typically involve adding a healthy copy of a gene into a patient’s cells to compensate for a defective one. These methods often rely on viral vectors to deliver genetic material and don’t directly alter the faulty DNA. While they can be effective, they may lead to unintended effects, limited control over where the gene inserts, and variable success rates.
CRISPR gene editing, on the other hand, works by directly targeting and modifying the DNA at the exact mutation site. Instead of simply adding genes, CRISPR can cut, delete, or repair faulty genetic sequences with high precision. This approach allows for permanent fixes at the root cause of genetic disorders. CRISPR is also faster, more affordable, and less invasive than many traditional methods, making it a game-changer in genetic medicine and personalized healthcare.
Top Genetic Diseases CRISPR Could Cure by 2030
CRISPR gene editing holds the potential to cure several inherited diseases by directly correcting the faulty DNA responsible for them. One of the most promising examples is sickle cell anemia, where clinical trials have already shown success in editing the defective gene that causes the disease. Beta-thalassemia, a similar blood disorder, is also being targeted with encouraging results. Another major focus is cystic fibrosis, a life-threatening lung disease caused by a single gene mutation that CRISPR may soon be able to repair at the molecular level.
Other genetic conditions in CRISPR’s sights include Huntington’s disease, muscular dystrophy, and even certain forms of inherited blindness. These diseases currently have no cures, but CRISPR offers the possibility of long-term or even permanent solutions. As research continues and safety improves, many of these therapies are expected to move from clinical trials to widespread use by 2030—redefining how we treat genetic disorders at their root.
CRISPR in Cancer Treatment: Promising Trials and Real Cases
CRISPR gene editing is showing powerful potential in the fight against cancer by helping scientists reprogram immune cells to better recognize and destroy tumor cells. One promising method involves editing T-cells, a type of white blood cell, to enhance their ability to attack specific cancer markers. In early clinical trials in the U.S. and China, CRISPR-edited T-cells have been safely infused into patients with leukemia, lymphoma, and lung cancer—showing signs of effectiveness and fewer side effects than traditional treatments.
Real cases from 2022 and 2023 have already demonstrated how CRISPR can be used to remove genes that limit T-cell activity, boosting the immune system’s power against cancer. For instance, a trial at the University of Pennsylvania successfully edited multiple genes in patient immune cells to treat resistant forms of blood cancer. While the technology is still under study, the results so far suggest that CRISPR gene editing could become a cornerstone of future cancer therapies—especially for hard-to-treat or relapsed cancers.
CRISPR and Personalized Medicine: Tailoring Treatments to Your DNA
CRISPR gene editing is revolutionizing personalized medicine by allowing treatments to be tailored to an individual’s unique genetic makeup. Unlike traditional “one-size-fits-all” approaches, personalized medicine uses genetic information to diagnose, prevent, and treat diseases more effectively. With CRISPR, doctors can potentially correct specific mutations in a patient’s DNA—offering highly targeted and lasting solutions for genetic conditions.
For example, if a patient has a rare genetic mutation causing a disorder, CRISPR can be programmed to precisely fix that error at the molecular level. This technology is especially promising for conditions like inherited cancers, rare blood diseases, and metabolic disorders. As research advances and genome sequencing becomes more accessible, CRISPR gene editing is expected to play a central role in customizing therapies—making medicine more accurate, efficient, and patient-centered by 2030.
Global CRISPR Research Leaders: USA’s Role in the Gene Editing Race
The United States is at the forefront of CRISPR gene editing research, leading the global race with cutting-edge innovations and clinical breakthroughs. Top institutions like Harvard University, MIT, and the Broad Institute have played a major role in developing and advancing CRISPR technologies since its discovery. In addition, companies like Editas Medicine, CRISPR Therapeutics, and Intellia Therapeutics, all based in the U.S., are pushing CRISPR therapies into real-world clinical use.
Federal support from agencies like the National Institutes of Health (NIH) and the FDA’s fast-track approvals for CRISPR-based trials have helped accelerate progress. While China and the EU are also making strides, the U.S. leads in both scientific output and investment in gene-editing startups. As global demand for genetic solutions rises, America’s leadership in CRISPR gene editing is expected to shape how these technologies are applied, regulated, and distributed worldwide by 2030.
The Ethical Debate: Where Should We Draw the Line on Human Gene Editing?
While CRISPR gene editing offers life-changing medical benefits, it also raises serious ethical questions—especially when it comes to editing the human genome. Many scientists agree that using CRISPR to treat life-threatening genetic diseases is justified. However, concerns arise when the technology is used for germline editing, where changes affect not just one individual but future generations. This includes controversial possibilities like enhancing intelligence, physical traits, or even designing babies.
Experts warn that without clear regulations and global consensus, CRISPR could lead to social inequality, genetic discrimination, or misuse for non-medical purposes. Ethical frameworks are being discussed in the U.S. and internationally to separate medical necessity from human enhancement. As CRISPR gene editing advances toward 2030, the challenge will be balancing scientific progress with moral responsibility—ensuring that this powerful tool is used to heal, not to divide.
CRISPR and Public Health: How It Could Reduce Global Disease Burden by 2030
CRISPR gene editing has the potential to dramatically reduce the global disease burden by 2030, especially in regions where access to traditional treatments is limited. By directly correcting the genetic causes of inherited conditions like sickle cell disease, thalassemia, and certain forms of blindness, CRISPR offers long-term or even permanent cures. Unlike conventional therapies, it targets the root cause of illness, reducing the need for lifelong medications or repeated procedures.
Beyond rare genetic disorders, CRISPR is also being explored to fight infectious diseases such as HIV and malaria. For example, scientists are testing gene-editing strategies to make immune cells resistant to HIV or to modify mosquitoes so they can’t spread malaria. As CRISPR becomes more affordable and accessible, it could be deployed globally to prevent disease at the population level—making it a powerful tool not just for individuals, but for improving overall public health outcomes.
The Cost of CRISPR: Will It Be Affordable by 2030?
CRISPR gene editing is currently one of the most advanced and promising technologies in medicine, but its high cost limits widespread access. Today, CRISPR-based treatments can range from $500,000 to over $2 million per patient, especially for rare genetic disorders. These prices reflect the complexity of development, clinical trials, and regulatory approval processes. As a result, only a small number of patients—mostly in developed countries—can benefit from these groundbreaking therapies.
However, by 2030, the cost of CRISPR gene editing is expected to decline significantly. Advancements in technology, mass production of gene-editing tools, and increased competition among biotech firms may drive prices down. In addition, insurance providers and public health systems are beginning to recognize the long-term savings of one-time genetic cures versus lifelong treatments. With the right policies and investment, CRISPR could become an affordable option for millions of patients around the world within the next decade.
What’s Next for CRISPR Gene Editing? 5 Bold Predictions for 2030
As CRISPR gene editing continues to evolve, experts foresee major breakthroughs that could reshape medicine and biology by 2030. First, we’re likely to see FDA-approved CRISPR treatments for common diseases like heart conditions, diabetes, and some cancers. Second, in vivo CRISPR therapies—where genes are edited directly inside the body—could become routine, making treatment faster and less invasive. Third, gene editing may expand into neurological diseases like Alzheimer’s and Parkinson’s, offering new hope for millions.
Fourth, CRISPR could help extend human healthspan by targeting aging-related genes, delaying the onset of age-related illness. Finally, in agriculture and public health, CRISPR may be used to eliminate disease-carrying insects or improve food resilience against climate change. With continued innovation, regulation, and investment, CRISPR gene editing is poised to move from labs to daily life—unlocking possibilities we are only beginning to imagine.
Real CRISPR Success Stories in 2025 You Should Know
CRISPR gene editing saw remarkable real-world progress in 2025, moving from experimental promise to tangible success stories that are transforming patient lives. One of the year’s biggest milestones was the FDA’s approval of a CRISPR-based therapy for sickle cell disease, marking the first time a gene editing treatment was cleared for wide clinical use in the United States. The therapy, developed by CRISPR Therapeutics and Vertex Pharmaceuticals, cured the condition in dozens of patients by permanently fixing the faulty hemoglobin gene.
Another major breakthrough involved beta-thalassemia, a serious blood disorder similar to sickle cell disease. In 2025, patients treated with CRISPR technology showed normal hemoglobin levels without needing regular blood transfusions—an outcome that was nearly impossible with traditional treatments. These successful cases prove that CRISPR gene editing is no longer theoretical but actively changing how genetic diseases are treated.
CRISPR also made strides in cancer treatment. At MD Anderson Cancer Center, researchers used gene-edited immune cells to shrink tumors in patients with aggressive leukemia and lymphoma. These personalized T-cell therapies, modified using CRISPR, targeted and destroyed cancer cells with minimal side effects, opening the door for new immunotherapy options.
Beyond individual diseases, CRISPR was used in 2025 to develop antiviral resistance, including early-stage work on HIV. Scientists at the University of Pennsylvania edited immune cells to resist infection by disabling genes that HIV uses to enter the cell. Though still in clinical trials, this success marks an important step toward long-term HIV treatment or prevention.
These real-world breakthroughs highlight how CRISPR gene editing is already reshaping medicine. As success stories continue to grow and access expands, 2025 will be remembered as a turning point when CRISPR moved from potential to practice—bringing hope to patients with conditions once thought incurable.
Conclusion: Why CRISPR Gene Editing Is the Future of Human Health
CRISPR gene editing is redefining the possibilities of modern medicine by offering precise, affordable, and lasting solutions to genetic diseases. Unlike traditional treatments that manage symptoms, CRISPR targets the root cause—our DNA—allowing scientists to correct mutations that lead to conditions like sickle cell disease, cystic fibrosis, and certain cancers. In 2025 alone, we’ve seen real success stories that prove this technology is no longer a future vision but a present-day reality. cdg
As we approach 2030, CRISPR is expected to become a key tool in personalized medicine, cancer therapy, global disease prevention, and even age-related health care. With ongoing advancements, reduced costs, and increased global access, this powerful technology holds the promise of transforming public health and individual treatment outcomes alike. Ethical oversight and responsible regulation will be critical, but with the right balance, CRISPR gene editing is set to become one of the most impactful innovations in human health history.
Subscribe to trusted news sites like USnewsSphere.com for continuous updates.





